Collaborators:
· Anna Duke
· Chinonso Ejere
Abstract: The problem to be determined in this lab is how the carbon dioxide and calcium carbonate affects the pH of water. The prediction was that the salt water would have a higher pH than the distilled water. The materials used are as follows: Two test tubes, universal indicator, ocean water, distilled water, balloon, plastic pipet. First, measure 10 mL of distilled ocean water into a test tube, and then add 10 mL of universal indicator and stir. Then, do the same thing but with ocean water instead of distilled. Then, blow up a balloon, secure the neck, and then cut the bulb of a plastic pipet off and put it in the neck of the balloon. Put the tip of the pipet into the ocean water sample, and release the carbon dioxide into the water. Do the same procedure for the distilled water. Lastly, add crushed calcium carbonate to both test tubes. After finishing the procedure, the results were as follows. The pH alone of the ocean water was 7, while the pH alone of the distilled water was six. When the carbon dioxide was added, the ocean water pH remained at 7, and the distilled water pH decreased to 5. When the calcium carbonate was added, both of the samples turned a lighter shade of green, but the distilled water was still a little darker and more red-ish than the ocean water.
Problem:
· How does carbon dioxide affect the pH of water?
Hypothesis: I predicted that the salt water would have a higher pH than the distilled water.
Experiment Parts:
· Control Group- Distilled water
· Experimental Group- Ocean water
· Independent Variables- Ocean water and distilled water
· Dependent Variable- pH
Materials:
· 2 test tubes
· Universal indicator
· Ocean water
· Distilled water
· Balloon
· Plastic pipet
Methods:
1. Measure 10 mL of ocean water into a test tube, and then add 10 mL of universal indicator to the tube, and stir.
2. Same as step #1, but use distilled water instead of salt water.
3. Blow up a balloon and seal it, then cut the bulb off of a plastic pipet and insert it into the neck of the balloon.
4. Release the balloon’s pressure into the ocean water sample, and record the results.
5. Release the balloon’s pressure into the distilled water sample, and record the results.
6. Add the crushed calcium carbonate into the ocean water and distilled water test tubes, and record the results.
Data:
· Anna Duke
· Chinonso Ejere
Abstract: The problem to be determined in this lab is how the carbon dioxide and calcium carbonate affects the pH of water. The prediction was that the salt water would have a higher pH than the distilled water. The materials used are as follows: Two test tubes, universal indicator, ocean water, distilled water, balloon, plastic pipet. First, measure 10 mL of distilled ocean water into a test tube, and then add 10 mL of universal indicator and stir. Then, do the same thing but with ocean water instead of distilled. Then, blow up a balloon, secure the neck, and then cut the bulb of a plastic pipet off and put it in the neck of the balloon. Put the tip of the pipet into the ocean water sample, and release the carbon dioxide into the water. Do the same procedure for the distilled water. Lastly, add crushed calcium carbonate to both test tubes. After finishing the procedure, the results were as follows. The pH alone of the ocean water was 7, while the pH alone of the distilled water was six. When the carbon dioxide was added, the ocean water pH remained at 7, and the distilled water pH decreased to 5. When the calcium carbonate was added, both of the samples turned a lighter shade of green, but the distilled water was still a little darker and more red-ish than the ocean water.
Problem:
· How does carbon dioxide affect the pH of water?
Hypothesis: I predicted that the salt water would have a higher pH than the distilled water.
Experiment Parts:
· Control Group- Distilled water
· Experimental Group- Ocean water
· Independent Variables- Ocean water and distilled water
· Dependent Variable- pH
Materials:
· 2 test tubes
· Universal indicator
· Ocean water
· Distilled water
· Balloon
· Plastic pipet
Methods:
1. Measure 10 mL of ocean water into a test tube, and then add 10 mL of universal indicator to the tube, and stir.
2. Same as step #1, but use distilled water instead of salt water.
3. Blow up a balloon and seal it, then cut the bulb off of a plastic pipet and insert it into the neck of the balloon.
4. Release the balloon’s pressure into the ocean water sample, and record the results.
5. Release the balloon’s pressure into the distilled water sample, and record the results.
6. Add the crushed calcium carbonate into the ocean water and distilled water test tubes, and record the results.
Data:
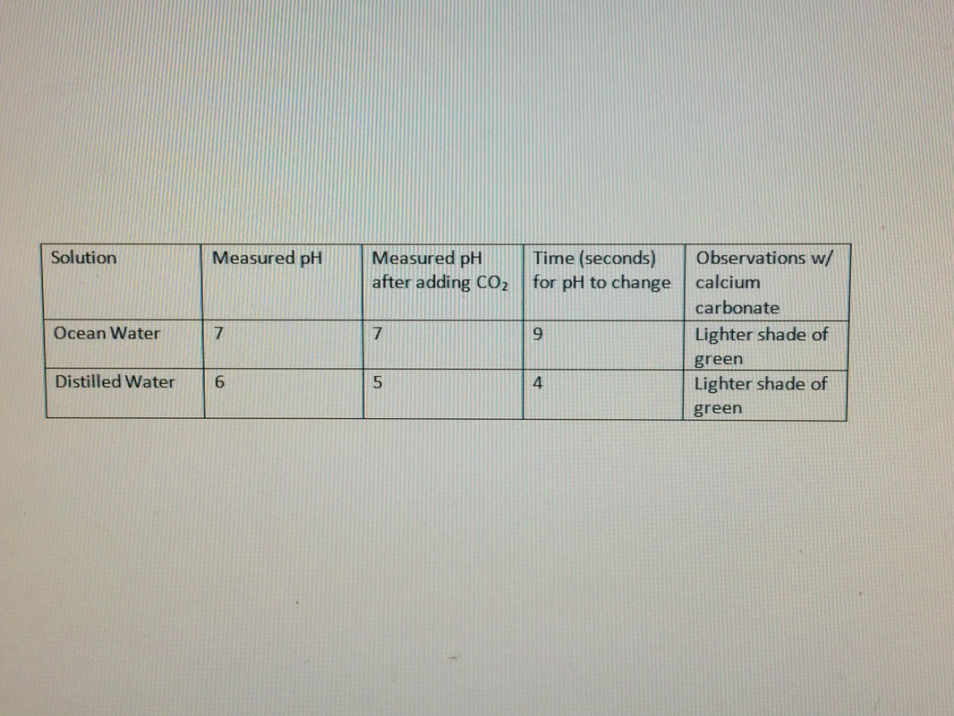
Data Table:
Data Analysis:
This data proves that the ocean water has a higher pH than the distilled water, proving the distilled water to be more acidic. The color of both of them after the carbon dioxide was added was different; the distilled was more red, while the ocean water sample was closer to green. However, after the calcium carbonate was added, both of them turned to a lighter shade of green. The distilled water was still a little darker than the ocean water though.
Conclusions:
The hypothesis that the ocean water would have a higher pH than the distilled water was correct. The pH for the ocean water after the calcium carbonate was 7, and in the distilled water it was 9. However, when the calcium carbonate was added to the ocean water, the pH remained the same while the distilled water’s pH decreased to 5, becoming more acidic. According to Ocean Acidification Network, carbonic acid is released when carbon dioxide dissolves into the water, and that releases the hydrogen ions. The more hydrogen ions there are, the higher the acidity. The most common pH of surface ocean water is 7. According to my source, ocean water has a higher pH. I got my pH as neutral. This might be because they tested a larger surface of water. We only used a small sample. When the carbon dioxide was added, the pH of the ocean water remained the same, while the pH of the distilled water decreased. The distilled water took much less time to react than the ocean water did. The distilled water took 4 seconds, while the ocean water took 9 seconds. I think this is because the distilled water sample was more acidic. When the calcium carbonate was added, both samples turned a lighter shade of green, but the distilled water was still slightly darker and more red-ish than the ocean water sample. I feel like this experiment is semi-accurate for testing the ocean absorption of carbon dioxide. Since the ocean is so big, it's hard to get completely accurate results since what's actually being tested is such a small sample. However, it's still partly accurate and can give a pretty good estimate.
Citation:
"Ocean Acidification Network." Ocean Acidification Network. Ed. Maria Hood, Dr., James Orr, Dr., Ken Caldeira, Dr., Richard Feely, Dr., and Scott Doney, Dr. N.p., n.d. Web. 03 Feb. 2014.
This data proves that the ocean water has a higher pH than the distilled water, proving the distilled water to be more acidic. The color of both of them after the carbon dioxide was added was different; the distilled was more red, while the ocean water sample was closer to green. However, after the calcium carbonate was added, both of them turned to a lighter shade of green. The distilled water was still a little darker than the ocean water though.
Conclusions:
The hypothesis that the ocean water would have a higher pH than the distilled water was correct. The pH for the ocean water after the calcium carbonate was 7, and in the distilled water it was 9. However, when the calcium carbonate was added to the ocean water, the pH remained the same while the distilled water’s pH decreased to 5, becoming more acidic. According to Ocean Acidification Network, carbonic acid is released when carbon dioxide dissolves into the water, and that releases the hydrogen ions. The more hydrogen ions there are, the higher the acidity. The most common pH of surface ocean water is 7. According to my source, ocean water has a higher pH. I got my pH as neutral. This might be because they tested a larger surface of water. We only used a small sample. When the carbon dioxide was added, the pH of the ocean water remained the same, while the pH of the distilled water decreased. The distilled water took much less time to react than the ocean water did. The distilled water took 4 seconds, while the ocean water took 9 seconds. I think this is because the distilled water sample was more acidic. When the calcium carbonate was added, both samples turned a lighter shade of green, but the distilled water was still slightly darker and more red-ish than the ocean water sample. I feel like this experiment is semi-accurate for testing the ocean absorption of carbon dioxide. Since the ocean is so big, it's hard to get completely accurate results since what's actually being tested is such a small sample. However, it's still partly accurate and can give a pretty good estimate.
Citation:
"Ocean Acidification Network." Ocean Acidification Network. Ed. Maria Hood, Dr., James Orr, Dr., Ken Caldeira, Dr., Richard Feely, Dr., and Scott Doney, Dr. N.p., n.d. Web. 03 Feb. 2014.